- Home
- Todays Market
- Indian Stock Market News May 2, 2017
Sensex Pares Early Gains; Pharma Stocks Drag Tue, 2 May 01:30 pm
After opening the day on a positive note, share markets in India pared early gains and are trading flat. Sectoral indices are trading on a mixed note with stocks in the realty sector and stocks in the oil and gas sector trading in green, while stocks in the pharma sector are leading the losses.
The BSE Sensex is trading up by 10 points (up 0.1%), and the NSE Nifty is trading up by 3 points (up 0.1%). Meanwhile, the BSE Mid Cap index is trading up by 0.1%, while the BSE Small Cap index is trading up by 0.2% The rupee is trading at 64.17 to the US$.
In news from stocks in the pharma sector. Lupin share price hit a fresh 52-week low in today's trade as The US Food and Drug Administration (USFDA) issued a form 483 to Lupin Ltd's Goa facility.
Form 483 relates to certain critical observations issued to a company at the end of an inspection if there were any violations of the Food Drug and Cosmetic Act and other related acts of the US Government.
Companies that receive its observations must respond in writing with a corrective action plan and implement it quickly. If the company does not meet the USFDA's expectations, a warning letter may be issued.
The USFDA cited three observations related to violation of good manufacturing practices following an inspection of the site between 27 March and 7 April this year.
The US drug regulator observed that the company failed to review and investigate unexplained discrepancy and out of specification components of a product batch at the facility.
The regulator also observed that Lupin did not have written procedures for production and process controls to assure quality of the product is as it is supposed to be as well as any established control procedures to monitor output of those manufacturing processes that cause variability in the characteristics of the drug.
On the face of it the observations seem to be strictly procedural in nature, and the company shall be able to resolve them in a few quarters at the most.
In the past, Lupin's Goa unit was inspected in July 2015 and February 2016. The July 2015's Form 483 was cleared in July 2016 and the latter in November 2016.
The Goa facility is an important unit for the company as its accounts for 30-40% of US sales. In the quarter ending in December 2016, Lupin's US sales grew 53.4% to US$ 316 million.
At the time of writing, Lupin share price was trading down by 1.5%.
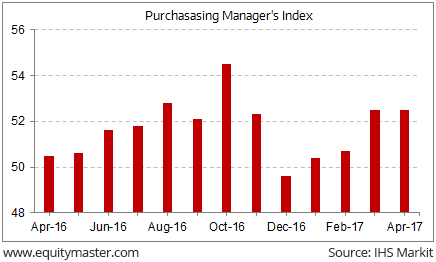
In news from India's manufacturing sector. The manufacturing sector continued its rebound from the notebandi induced downturn. Indian manufacturing activity expanded for a fourth consecutive month in April, as buoyant domestic demand coupled with sustained growth of new orders from abroad boosted the upturn in total new business received by Indian manufacturers in April, according to the Nikkei Purchasing Managers' Index (PMI) survey by Markit.
The PMI is the reading of the country's manufacturing sector output and is updated monthly. A reading above 50 indicates expansion, while any score below the mark denotes contraction.
Having deteriorated in December for the first time in one year, the health of India's manufacturing economy showed signs of improvement in January 2017. The manufacturing PMI recovered from 49.6 in December 2016, to 50.4 in January 2017 and continued moving upwards marginally to 50.7 in February.
The sector continued its upward move, as PMI rose to 52.5 in March, registering the fastest upward move since October 2016. At 52.5 in April, the PMI remains unchanged from, yet signifying expansion in the manufacturing sector.
PMI Continues Expansion
It is evident from the above chart that the manufacturing activity is inching towards the pre-demonetisation levels noted in October 2016. However, manufacturing is seen to steadily pick up from the notebandi blues.
The increase in demand only provided a more modest lift to overall output and employment as higher prices of raw materials ate into firms' profits.
But despite rising cost burdens, factory gate prices barely rose as owners tried to sustain demand still recovering from Prime Minister Narendra Modi's shock move late last year to ban high-value currency notes that caused a huge disruption.
Decent economic growth and low inflation support the Reserve Bank of India's recent shift to neutral from an accommodative policy stance.
Looking ahead, production volumes are likely to rise further as businesses will seek to replenish their stocks, as remonetisation nears completion.
For information on how to pick stocks that have the potential to deliver big returns, download our special report now!
Read the latest Market Commentary


Equitymaster requests your view! Post a comment on "Sensex Pares Early Gains; Pharma Stocks Drag". Click here!
Comments are moderated by Equitymaster, in accordance with the Terms of Use, and may not appear
on this article until they have been reviewed and deemed appropriate for posting.
In the meantime, you may want to share this article with your friends!