- Home
- Todays Market
- Indian Stock Market News August 2, 2017
Sensex Trades in Red; FMCG Stocks Witness Selling Wed, 2 Aug 01:30 pm
After opening the day marginally higher, share markets in India witnessed cautious trading activity eyeing the RBI monetary policy decision scheduled today, and are presently trading marginally below the dotted line. Sectoral indices are trading on a mixed note with stocks in the power sector and consumer durables sector witnessing maximum buying interest. Metals stocks and FMCG sector are trading in the red.
The BSE Sensex is trading down by 50 points (down 0.2%) and the NSE Nifty is trading down by 24 points (down 0.2%). The BSE Mid Cap index is trading down by 0.3%, while the BSE Small Cap index is trading up by 0.2%. The rupee is trading at 63.91 to the US$.
In news from stocks in the pharma sector, Lupin is in focus today after the pharma major received US Food and Drug Administration (USFDA) approval to market Fluocinonide Topical ointment, an anti-inflammatory drug, in the American market.
The ointment is a generic version of County Line Pharmaceuticals' Lidex ointment and is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Citing IMS MAT sales data, the company said that Lidex had sales of US$ 40.3 million in the US market till March 2017.
Lupin has so far received approval for 220 products and is still awaiting USFDA nods for 148 more products.
Recently, the drugmaker's Aurangabad manufacturing plant underwent USFDA inspection. During the inspection, the US Food and Drug administration (USFDA) issued one 483 observation. The company said that the observation was procedural and would be corrected during the inspection itself.
As per USFDA, observations are made in Form 483 when investigators feel that conditions or practices in the facility are such that products may become adulterated or render injuries to health.
In recent times, pharma companies have been bogged down by mounting pressure from US Food and Drug Administration (USFDA) to adhere to quality standards at their manufacturing plants. In the past three years, the USFDA raised numerous regulatory concerns resulting in import bans and suspension of new drug approvals from facilities of Indian pharma companies.
However, new drug approvals are seen to pick up sharply in 2017.
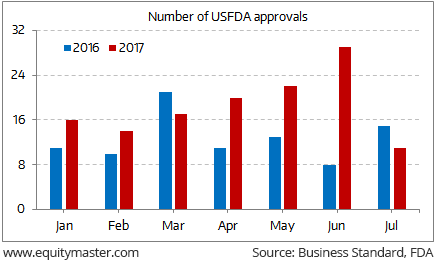
USFDA sweetener for Indian Pharma
During the period January-July 2017, 129 approvals for generic drugs were made. This is 45% higher from 89 approvals made in the corresponding period last year.
As per the Indian Pharmaceutical Alliance, the pace of drug approvals has gained momentum after they complained to FDA about delays last year. Approvals for drugs have also picked up after USFDA concerns at some of the manufacturing units were addressed.
The USFDA's move to speed up generic drug approvals in a bid to reduce healthcare costs is likely to provide some earnings relief to domestic pharma companies.
At the time of writing, Lupin share price was trading up by 2.5%.
Moving on to news from stocks in the FMCG sector. Marico opened the day lower by 5% today after it posted tepid results for the June quarter.
Marico reported a 12% fall in the quarter's net profit at Rs 2.36 billion as against Rs 2.68 billion posted in the same period last year.
The FMCG major saw a 4% drop in its consolidated revenue, owing mostly due to a dip in its volumes, which were down by over 9% in the quarter.
The company attributed the volume fall to de-stocking effect in June due to Goods and Services Tax (GST). The company also said that the decline is attributable to steep pipeline correction.
The company further said that it held back price increase in Parachute rigid portfolio due to GST rollout.
At the time of writing, Marico share price was trading down by 1.8%.
On the corporate earnings front, the GST implementation is said to impact near term earnings of the companies. However, over a long run, expectations are that earnings would normalize. In addition, GST is expected to be a big support to the depressed earnings of the organised listed Indian companies in the long run.
However, GST is a complicated topic, the exact implications of which will be obvious over next few years. Vivek Kaul, editor of Vivek Kaul's Diary has studied the finer aspects of GST and predicted what could go right and wrong.
To know more, download his special report - The Good, the Sad and the Terrible (GST)
For information on how to pick stocks that have the potential to deliver big returns, download our special report now!
Read the latest Market Commentary


Equitymaster requests your view! Post a comment on "Sensex Trades in Red; FMCG Stocks Witness Selling". Click here!
Comments are moderated by Equitymaster, in accordance with the Terms of Use, and may not appear
on this article until they have been reviewed and deemed appropriate for posting.
In the meantime, you may want to share this article with your friends!